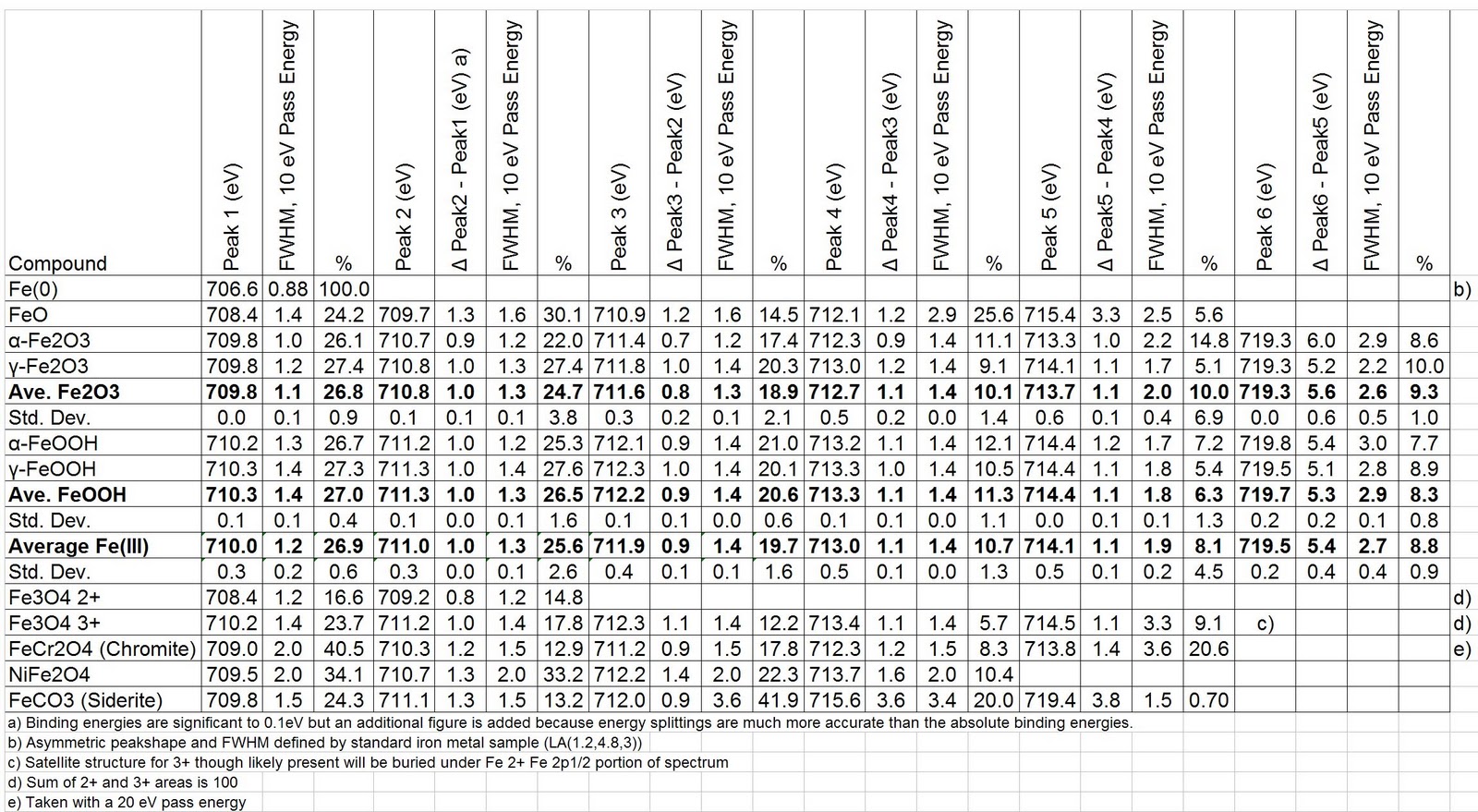
The experimentally determined Curie constants from the susceptibility data are in good agreement with the calculated values based on the Fe3+, Fe2+ and V4+ concentrations determined from the XPS analyses. The measured Fe 2p XPS spectra were analyzed using a simplified peak model with the main objective to quantify the relative proportions of metallic and oxidic iron and thus only the Fe 2p 3/2 peak was considered in the fit (for details, see the Supporting Information, Section 3.3). The magnetic susceptibility data appear to follow a Curie–Weiss behavior (χ=C/(T−θ)) for temperatures above ∼15 K with negative paramagnetic Curie temperatures indicative of antiferromagnetic interactions between the magnetic Fe3+, Fe2+ and V4+ ions. In addition, the broadened V 2p3/2 and Fe 2p3/2 spectra for the glass samples are decomposed into two peaks that are associated with the presence of V5+ and V4+ and Fe3+ and Fe2+, respectively. The O 1s spectra for the glasses are single, symmetric peaks arising from nearly 95% of the oxygen sites being occupied by non-bridging oxygen atoms. The core level binding energies of V 2p, Fe 2p, P 2p and P 2s in the Fe2O3–vanadium phosphate glasses have been measured and shifts from the corresponding binding energies measured for V2O5, Fe2O3 and P2O5 powders are accounted for by changes in the next-nearest neighbor environment of these atoms in the P4O10 local structure. Samples of the iron oxides Fe 0.94 O, Fe 3 O 4, Fe 2 O 3, and Fe 2 SiO 4 were prepared by high temperature equilibration in controlled gas atmospheres. Vanadium phosphate glasses containing Fe2O3 with the chemical composition, where x=0.00, 0.10, 0.20, 0.25 and 0.30, have been prepared and investigated by X-ray photoelectron spectroscopy (XPS) and magnetic susceptibility measurements.
